Fungi
Introduction and context
The primary task of the mycology lab is to perform diagnoses on plants and plant products that are suspected to be infected with a plant-pathogenic fungus. We also detect specific fungi such as Verticillium, Pythium and Phytophthora in soil and water samples. Upon request, fungal isolates can be determined via morphological characteristics or via sequencing of specific genes (rDNA ITS, beta-tubuline, COX, etc.) and aligning the sequences with those in the databases.
We have been assigned by the FASFC to perform accredited (ISO 17025) and non-accredited analyses for the detection of quarantine fungi in the context of national detection surveys and/or import or export monitoring of plants and plant-related products. The majority of such analyses concern Phytophthora ramorum, P. kernoviae, Monilinia fructicola, Tilletia indica, Claviceps purpurea en Guignardia citricarpa.
In addition to detection and diagnosis, we also perform resistance assays. We have expertise in determining the susceptibility of candidate chrysanthemum cultivars to pathotypes of Puccinia horiana. We also contribute to the screening of the susceptibility of maize cultivars to Rhizoctonia solani. Upon request and in collaboration with our clients, we can also develop and perform other bioassays with other organisms.
This lab is actively involved in the working of the European Mycological Network (EMN), through which we have access to international expertise and the latest protocols. In turn, the European and Mediterranean Plant Protection Organisation (EPPO) uses EMN as an expert body for development and evaluation of protocols for regulated fungi.
Diagnosis and detection procedures?
The analysis used depends on the type of pathogen and the symptoms presented.
Obligate pathogens (such as false or real powdery mildew) are determined directly on the plant. This is generally done using microscopic analysis of the fungal structures. Incubating the plant under humid conditions is sometimes required.
Non-obligate pathogens are generally identified after isolation and plating on various culture media (PDA, P5ARP, WA, Komada’s, etc.). In addition, the samples are generally incubated for possible direct study.
 | 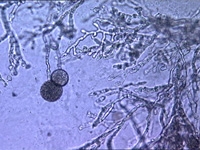 | 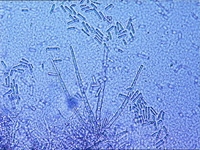 |
| Microscopic identification | mycelium with spores | spores Thielaviopsis basicola |
Detection of pathogenic fungi in water samples is done either directly via filtration and plating of the filter on culture media, or indirectly using a rhododendron leaf bait assay. The direct method is based on the direct isolation of the living fungal fragment from the water and is thus quantitative. This method is usually used to check whether the disinfection system for fertilisation systems in hydroculture (UV, slow sand-filtering, etc.) is working properly.
The leaf bait test is indirect and is therefore only semi-quantitative but this method is more appropriate for use in the sensitive detection of Pythium or Phytophthora in large volumes such as storage tanks. For this bait test, the leaves are suspended in a water tank during 3-5 days. In that time the zoospores of Pythium or Phytophthora can infect the leaf. The leaves are sent to the lab where the lesions are analysed using plating and microscopic determination of the outgrowing Pythium and Phytophthora species.
To detect soil pathogens, bait tests are used (e.g. for Phytophthora species) or extraction techniques (e.g. for the microsclerotes of Verticillium dahliae). Such techniques allow a relatively large and thus representative sample to be tested. These analyses usually are done in combination with plating on semi-selective media such as MSEA.
 |  |
| shaking sieving apparatus | microsclerotes |
In specific cases, the plant samples, water samples and soil samples are also tested using molecular techniques. These methods are based on DNA extraction and classical or real-time PCR. They give a relatively fast and reliable indication of the presence or absence of a target organism.
In cases where no plant pathogenic organism can be shown as the cause of damage, we try to determine what another possible cause could be. Growth problems can be caused by the wrong substrate or the use of herbicides. Techniques such as pH and conductivity measurements or bioassays using garden cress (Lepidium sativum) are then used.
 Diagnostic Centre for Plants
Diagnostic Centre for Plants